  Use the following information to prepare the September cashīudget for PTO Manufacturing Co. Rank the following elements by increasing atomic radius: carbon, aluminum, oxygen, potassium.There is a 250-m-high bluff at Half Dome in Yosemite Nationalįoxwood Company is a metal and woodcutting manufacturer, sellingĬorning Company has a decentralized organization with a 7.48 Arrange the elements in each of the following groups in order of increasing.Consider the titration of 50.0 mL of 0.133 M NH3 (a weak base.X and output y: (a) sketchbthe input.ĭynamic analysis of a lifting task be higher or lower. (Convolution DT) consider the following LTI system with input. 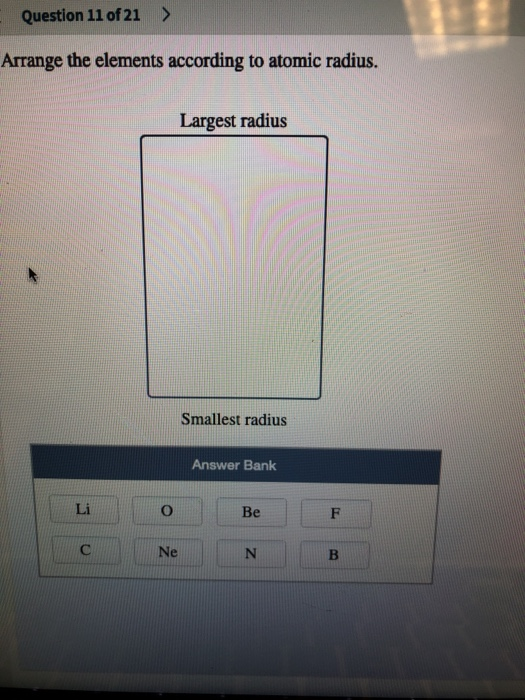
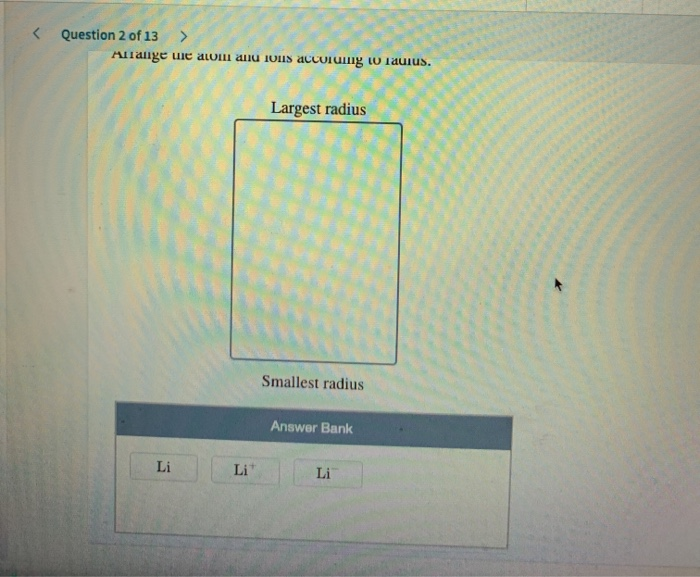
Suppose you conduct a hypothesis test to determine. Use the following information for questions 37-38: Variables with means µx and µy respectively. Let X and Y be independent identically distributed random Figure 8.5 Comparisons of the radii, in, of neutral atoms and ions for several of the groups of representative elements. Arrange the elements according to atomic radius. series#a) (1 mark) Arrange these elements in order of increasing atomic radius, from smallest to largest: Sb, S, Pb, Se Smallest smallest b) (1 mark) Arrange this isoelectronic series in order of increasing radius, from smallest to largest: Se?, Sr2*, Rb, Br Smallest Largest c) (1 mark) Arrange these elements in order of increasing first ionization energy, from smallest to largest: CI, S, Ge, Pb smallest < < < Largest NAME AND STUDENT NUMBER: 2. a) (1 mark) Arrange these elements in order of increasing atomic radius, from smallest to.ġ. The largest bank of answers Oxygen Silicon Calcium Barium. To rank items as equivalent, overlap them. Arrange the elements according to atomic radius, from largest to smallest. A carbon-12 atom is made up of six Trc nghim T vng Ting Anh lp 12. Place the following elements in order of decreasing atomic size: lead, phosphorus, oxygen, cesium, barium, and silicon Rank from largest to smallest. Pursuant to the 2015 Budget Act, as of July 1, 2015, CalHR only submits to the. So carbon has fewer energy levels than aluminum, silicon and that is the predominant factors. So, uh, when you go down, you get more energy levels and that makes the atoms bigger.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
